 On the Case
On the Case
By Atul Kumar, MD, MS; Ryan Joyce, MD; and Marco Molina, MD
Radiology Today
Vol. 20 No. 3 P. 30
History
A 52-year-old woman with a past medical history of high-grade endometrial carcinoma and status post hysterectomy and bilateral salpingo-oophorectomy presented with concerns of a postoperative umbilical hernia. To assist the surgeon with preoperative planning for the presumed umbilical hernia, an IV contrast-enhanced abdominal and pelvic CT was obtained.
Findings
Axial and reconstructed coronal IV contrast-enhanced CT images revealed a 2.3 cm saccular aneurysm arising from the anterior margin of the origin of the main portal vein, just posterior to the pancreatic neck (Figures 1 and 2). Upon review of prior imaging reports, this aneurysm has remained stable in size for three years.
Diagnosis
Portal venous system aneurysm.
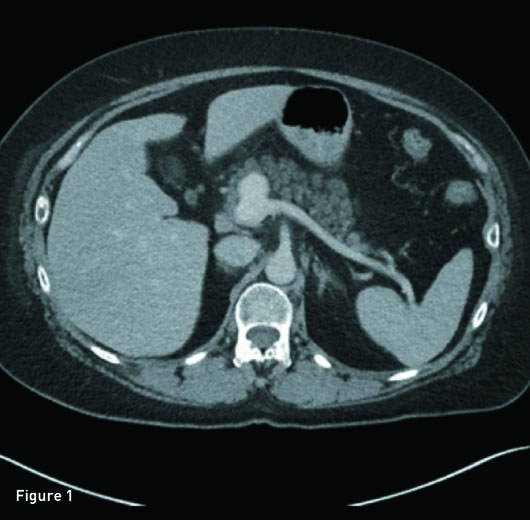
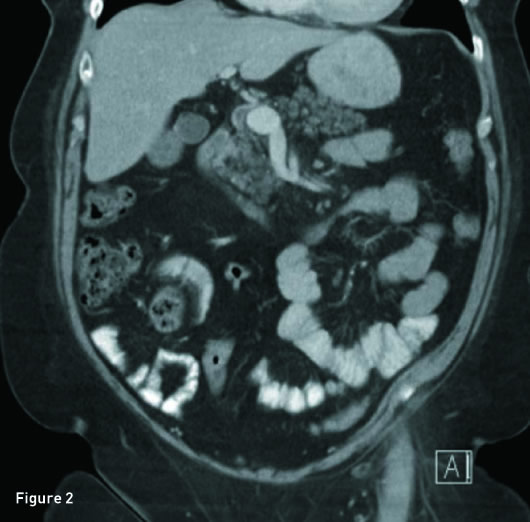
Discussion
A portal vein aneurysm is defined as a focal fusiform or saccular dilatation within the portal system. The first case was described as early as 1956 by Barzilai and Kleckner. Portal vein system aneurysms are considered rare, as they make up only 3% of all venous aneurysms. Today, they are discovered more frequently as incidental findings due to the increased use of imaging.
The extrahepatic portal vein is considered normal in size if the diameter is ≤15 mm, while in cirrhotic patients it measures ≤19 mm. It is aneurysmal (fusiform) when the diameter is ≥20 mm. For an intrahepatic portal vein fusiform aneurysm, it is considered aneurysmal if the diameter is ≥7 mm in normal patients and ≥8.5 mm in cirrhotic patients. Saccular aneurysms are diagnosed by the presence of a focal vessel wall outpouching/morphology and do not have to meet specific size criteria for diagnosis.
Portal vein aneurysms are most commonly found at bifurcations and confluences of the portal venous system such as the superior mesenteric vein/splenic vein confluence, main portal vein, and intrahepatic portal vein bifurcation sites.
The pathogenesis of portal vein aneurysms is thought to be either congenital or acquired. The congenital etiology is thought to occur either due to incomplete regression of the distal right vitelline vein leading to a focal diverticulum, which forms into an aneurysm or an inherent weakness in the wall of the portal system during organogenesis. The most common acquired cause is portal hypertension secondary to cirrhosis or chronic liver disease resulting in high-pressure splanchnic flow leading to a weakening of the vascular wall. Other acquired causes include necrotizing pancreatitis, trauma, abdominal surgery, or invasion of the portal vein by malignancy.
Clinically, one-third of patients are asymptomatic upon presentation, and the portal vein aneurysm is found incidentally on imaging. Approximately 50% of patients with a portal vein aneurysm present with nonspecific abdominal pain.
A low risk of complications is observed, the most common being thrombus formation within the aneurysm. Other complications that have been identified include aneurysm rupture, inferior vena cava obstruction, duodenal compression, or common bile duct obstruction.
On ultrasound imaging, portal vein aneurysms present as anechoic cyst-like lesions in the region of the porta hepatis but can also be echogenic due to the presence of mural thrombus. Color or Doppler sonography can be used to show blood flow with a nonpulsatile monophasic waveform. CT or MRI can also be used, especially for surgical planning or equivocal ultrasound findings.
Treatment of portal vein aneurysms is not standardized. No clear evidence or recommendations exist on treatment guidelines, as with other types of aneurysms such as abdominal aortic aneurysms. Conservative management is often considered as aneurysms are often stable and only require surveillance imaging, especially in asymptomatic patients who have no risk factors such as portal hypertension or liver disease. An increase in size can indicate portal hypertension and warrant further workup and possible surgical intervention as rupture can lead to death. Surgical management should also be considered if a patient is symptomatic or the aneurysm is thrombosed.
— Atul Kumar, MD, MS, is a radiology resident at UConn Health, University of Connecticut.
— Ryan Joyce, MD, is a radiology resident at UConn Health, University of Connecticut.
— Marco Molina, MD, is the section head for body imaging and program director of diagnostic radiology at UConn Health, University of Connecticut.
Resources
1. Barzilai R, Kleckner MS Jr. Hemocholecyst following ruptured aneurysm of portal vein; report of a case. AMA Arch Surgery. 1956;72(4):725-727.
2. Doust BD, Pearce JD. Gray-scale ultrasonic properties of the normal and inflamed pancreas. Radiology. 1976;120(3):653-657.
3. Tolgonay G, Ozbek SS, Oniz H, Süzer E, Yurdakul LO. Regression of splenic vein aneurysm following resolution of splenomegaly. J Clin Ultrasound. 1998;26(2):98-102.
4. Gallego C, Velasco M, Marcuello P, Tejedor D, De Campo L, Friera A. Congenital and acquired anomalies of the portal venous system. Radiographics. 2002;22(1):141-159.
5. Labgaa I, Lachenal Y, Allemann P, Demartines N, Schäfer M. Giant extra-hepatic thrombosed portal vein aneurysm: a case report and review of the literature. World J Emerg Surg. 2014;9:35.
6. Rafiq SA, Sitrin MD. Portal vein aneurysm: case report and review of the literature. Gastroenterol Hepatol (N Y). 2007;3(4):296-298.
7. Laurenzi A, Ettorre GM, Lionetti R, Meniconi RL, Colasanti M, Vennarecci G. Portal vein aneurysm: what to know. Dig Liver Dis. 2015;47(11):918-923.
8. Koc Z, Oguzkurt L, Ulusan S. Portal venous system aneurysms: imaging, clinical findings, and a possible new etiologic factor. AJR Am J Roentgenol. 2007;189(5):1023-1030.

