 On the Case
On the Case
By Ved Caculo, MBBS, MD; Swamini Panandiker, MBBS, DMRD, DNB; Marizela J. D. C. Carvalho, MBBS; and Shikha Lawande, MBBS
Radiology Today
Vol. 22 No. 4 P. 30
History
A 15-day-old male infant, born at term to a 31-year-old gravida 2 para 1 woman by spontaneous vaginal delivery, presented with lethargy and failure to thrive because of inadequate breast-feeding. Other symptoms included oliguria with high-colored urine, reduced activity, and alteration in the level of consciousness for four days. Antenatal history was uneventful. There was no asphyxia at birth. The baby had a birth weight of 3.65 kg. Immediately after birth, there was no neonatal hyperbilirubinemia, hypoglycemia, or feeding intolerance.
On physical examination, the baby’s weight, height, and head circumference were 2.58 kg (a decrease of 30%), 49 cm, and 34.5 cm, respectively. The baby had doughy skin and reduced skin turgor. Hypotonia with poor activity was also noted. The baby also had dry mucous membranes with a sunken fontanelle.
The blood level of urea was 420 mg, serum creatinine 4 mg, sodium 198 mmol/L, potassium 70 mmol/L, calcium 8.8 mmol/L, albumin 2.1 mg, and alkaline phosphatase 109 IU/L. The arterial blood pH was 7.3 and bicarbonate was 12.3, pCO2 25. The prothrombin time was 22 seconds with an international normalized ratio of 2.1 and a partial thromboplastin time >60. The baby’s hemoglobin was 18.4 mg with a total count of 23,710 showing a differential count of 50% neutrophils and 41% lymphocytes. Platelet count was 1.1 lakh, or 110,000 IU.
A working diagnosis of hypernatremic dehydration with acute renal failure was made based on the clinical and laboratory parameters. Bolus IV fluids were started, followed by maintenance fluids with breast-feeding. Subsequently, the baby went into fluid overload. Hence, a short course of furosemide was given. During the course of the illness, the baby developed hyperglycemia and hypokalemia. Peritoneal dialysis, potassium correction, and insulin infusion were administered.
A renal ultrasound was performed. The baby eventually showed signs of progressive deterioration of sensorium. Noncontrast CT of the brain revealed features suggestive of hypoxic-ischemic injury. The baby also developed respiratory failure and was put on ventilatory support (synchronized intermittent mandatory ventilation plus pressure-controlled ventilation) with the following parameters: peak inspiratory pressure: 18; positive-end expiratory pressure: 6; beats per minute: 40; and fraction of inspired oxygen: 40%. The baby expired on the 39th day of life.
Findings
On renal ultrasound, the right kidney measured 4.5 cm X 2.5 cm and the left kidney measured 4.3 cm X 2.6 cm. Diffusely increased echogenicity involving the base of the renal pyramids was noted bilaterally with these echogenic foci measuring approximately 4 to 5 mm and were suggestive of medullary nephrocalcinosis. Renal cortical caliber was normal bilaterally. There was bilateral dilatation of pelvicalyceal system with anteroposterior diameter of the renal pelvis measuring 11 mm on the right and 9 mm on the left (Figures 1 to 4). There was moderate left hydroureter and no evidence of hydroureter on the right side. The renal pelvic dilatation was consistent with bilateral ureteropelvic junction obstruction, which needed interval follow-up prior to intervention. No urinary bladder abnormality was noted.
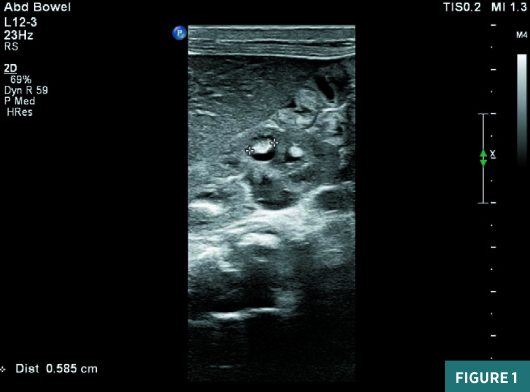
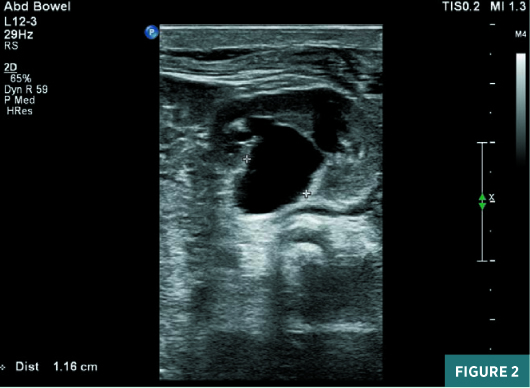
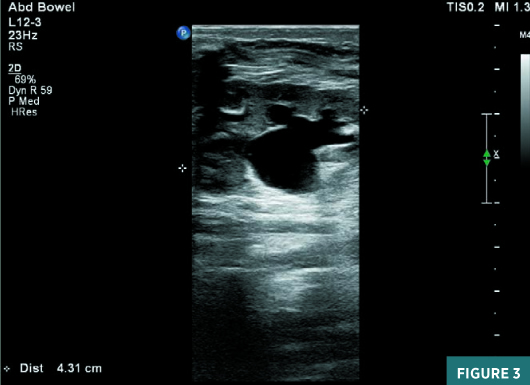
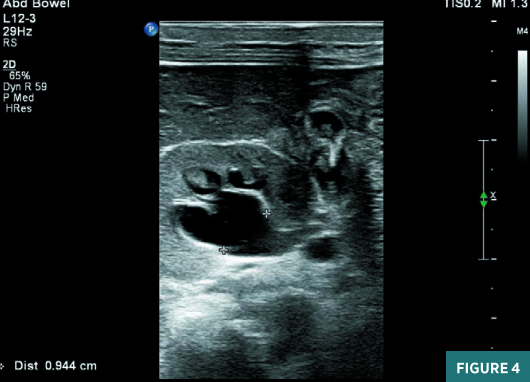
Diagnosis
Neonatal medullary nephrocalcinosis.
Discussion
Nephrocalcinosis is the deposition of calcium in the kidney parenchyma. It includes deposition of calcium phosphate and calcium oxalate. It is subdivided into medullary or cortical nephrocalcinosis.1
Nephrocalcinosis occurs more frequently in neonates, especially preterm infants, because of renal tubular immaturity, which leads to enhanced crystal formation and aggregation, particularly in the setting of tubular fluid supersaturation, which is promoted by hypercalciuria and hypocitraturia.2 Other common causes of nephrocalcinosis in children include medications including loop diuretics such as furosemide, methylxanthines, glucocorticoids, and vitamin D (in pharmacological doses), as well as renal tubular acidosis, idiopathic hypercalciuria, hyperparathyroidism, and hyperoxaluria (associated with parenteral nutrition). Bartter syndrome, pseudo-aldosteronism, hyperphosphaturia, and conditions causing hypercalcemia may also be associated with nephrocalcinosis.3
Nephrocalcinosis secondary to primary hyperoxaluria/oxalosis, renal tubular acidosis, or urinary tract infections is rare in newborns, although these conditions may present a few months after birth.4 On ultrasonography, in patients with nephrocalcinosis, the renal pyramids are echogenic and may or may not have associated shadowing, which depends on the size of the calcifications. The size, echogenicity, and pattern of the medullary echogenic foci point toward medullary nephrocalcinosis. In individual cases, it can be difficult to distinguish between medullary nephrocalcinosis and renal calyx calculi.
In this case, the medullary nephrocalcinosis may have been secondary to hypernatremia or was furosemide induced. Factors that predispose neonates to medullary nephrocalcinosis need focused imaging in neonates and infants with deranged metabolites or signs of acute kidney injury. These foci of medullary hyperechogenicity need to be differentiated from Tamm-Horsfall proteins, which are physiological in the neonatal period. Hypernatremia-induced medullary nephrocalcinosis is reversible and needs follow-up focused renal ultrasonography after treatment.5
— Ved Caculo, MBBS, MD, is a senior resident in the department of radiodiagnosis at Goa Medical College in Bambolim-Goa, India.
— Swamini Panandiker, MBBS, DMRD, DNB, is a lecturer in the department of radiodiagnosis at Goa Medical College.
— Marizela J. D. C. Carvalho, MBBS, is a resident in the department of radiodiagnosis at Goa Medical College.
— Shikha Lawande, MBBS, is a resident in the department of radiodiagnosis at Goa Medical College.
References
1. Adam A, Dixon AK, Gillard JH, Schaefer-Prokop C, eds. Grainger & Allison’s Diagnostic Radiology. 7th ed. Amsterdam, Netherlands: Elsevier; 2020.
2. Smith J, Stapleton FB. Nephrocalcinosis in neonates. UpToDate website. https://www.uptodate.com/contents/nephrocalcinosis-in-neonates. Updated March 2021.
3. Lodha R, Hari P, Bagga A. Syndrome of renal magnesium wasting and nephrocalcinosis. Indian Pediatr. 1999;36(10):1046-1048.
4. Eichenwald EC, Hansen AR, Martin CR, Stark AR, eds. Cloherty and Stark’s Manual of Neonatal Care. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2016.
5. Ali US, Sengupta K, Andankar P, Saraf S, Chawla A, Deshpande S. Reversible renal medullary hyperechogenicity in neonatal hypernatremic dehydration. Pediatr Nephrol. 2004;19(9):1050-1052.

