 On the Case
On the Case
By Brad Kincaid, MS, MD; Nicholas Arconti, BA; and Mark Kane, MD
Radiology Today
Vol. 24 No. 3 P. 30
History
A 79-year-old male with a history of gait instability and atrial fibrillation treated with anticoagulants presented to the emergency department after a mechanical fall onto a hard tile floor while exercising. Physical exam revealed blunt soft tissue injury and abrasions to the right forehead with soft tissue swelling. Initial head CT reported a small, right frontal scalp hematoma without significant intracranial injury. Follow-up CT performed 10 days later revealed decreased soft tissue swelling at the area of injury, however, with increasing size of a focal hematoma.
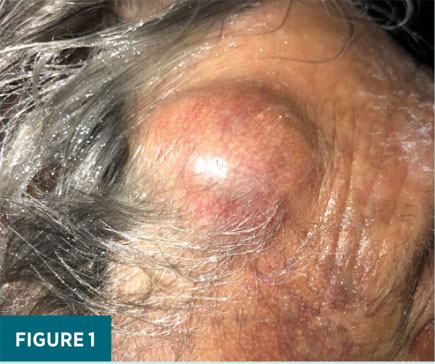
Three weeks after his injury, the patient went to the dermatologist for evaluation of a right scalp mass that was progressively enlarging. Clinical examination by the dermatologist revealed a compressible mass with ecchymosis that was mildly tender to palpation, measuring 4 cm in diameter by 2 cm in height (Figure 1). For further interrogation, a duplex ultrasound was ordered to assess for active bleeding.
Findings
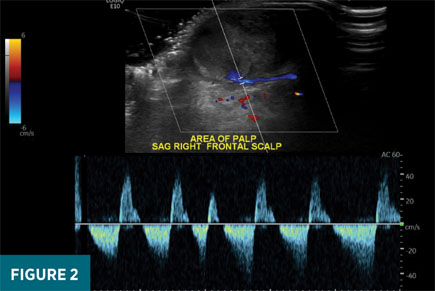
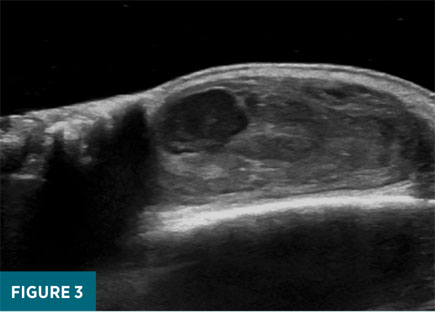
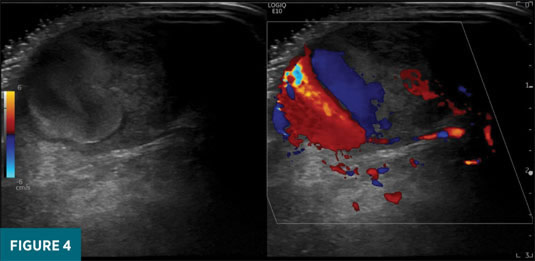
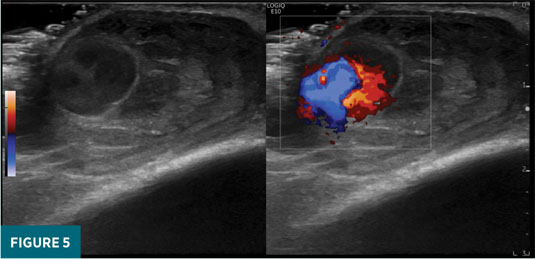
Initial duplex ultrasound examination of the right frontal scalp revealed a 1.6 X 2.7 X 2.7 cm well-circumscribed, heterogeneously echogenic mass just deep to the dermis (Figure 2). Grayscale images revealed turbulent pulsatile motion within the lateral aspect of the mass, and color Doppler interrogation revealed a “see-sawing” region of internal Doppler flow measuring 1.2 X 1.0 cm (Figures 3-5). These findings were consistent with a pseudoaneurysm of the right frontal scalp with a component of solid hematoma and an actively bleeding region represented by the area of see-sawing Doppler flow. The patient was referred to vascular surgery, and his apixaban therapy was halted. After discussion with the surgeon, a plan was made to follow up with duplex ultrasound in one week to assess for interval change. Ultrasound performed six days later revealed an approximately stable-in-size (1.4 X 2.2 X 2.8 cm) hematoma with an interval decrease in the region of internal Doppler flow (0.5 X 0.6 cm compared with 1.2 X 1 cm). The patient remained off anticoagulation and a final follow-up ultrasound performed 19 days later revealed an interval decrease in maximal size of the pseudoaneurysm to 2.1 cm (previously 2.8 cm) with lack of internal color Doppler flow compatible with complete thrombosis.
Diagnosis
Posttraumatic Superficial Temporal Artery (STA) Pseudoaneurysm
Discussion
STA pseudoaneurysm is a rare diagnosis, with only a few hundred cases reported in the literature.1 The diagnosis can be made based on physical exam and history, which typically reveals a “pulsatile preauricular mass” after recent injury to the frontotemporal region.2 In equivocal cases, CT angiography or ultrasound examination can also provide diagnostic support.
The STA, along with the maxillary artery, is one of the two terminal branches of the external carotid artery. It arises within the parotid gland, from which it courses superficially to the posterior aspect of the zygomatic process of the temporal bone before bifurcating into a parietal and frontal branch, usually at the level of the zygomatic arch. Of the two branches, the frontal branch is typically more prone to traumatic injury, as, throughout its course, it is either directly adjacent to the skull or only minimally protected by the relatively thin temporalis and frontalis muscles.3 Pathophysiologically, a pseudoaneurysm occurs when an arterial wall defect results in development of a hematoma that remains in communication with the arterial lumen, resulting in persistent blood flow to and from the extra-arterial site. The vast majority of STA pseudoaneurysms are a result of blunt trauma ranging from sports related injuries to assault or motor vehicle accidents, with a few rare cases occurring after iatrogenic or penetrating injury.4
Although rare—only a few hundred cases have been reported in the literature— the typical clinical course of a posttraumatic STA pseudoaneurysm has been defined by a relatively delayed development of a progressively enlarging, preauricular, pulsatile mass that usually becomes noticeable two to six weeks after the inciting injury.5 Clinically, patients tend to complain of a painless, pulsating sensation over the area of the lesion, although some patients may complain of an accompanying throbbing headache. On physical exam, a systolic pulsation and/or thrill may be appreciated, differentiating a pseudoaneurysm from an arterio-venous fistula, which will present with a continuous thrill. The degree of visible pulsation is dependent on intralesional blood flow; a completely thrombosed pseudoaneurysm will lack the classic pulsating waveform, broadening the differential diagnosis to include more common entities, including arteriovenous fistula, lipoma, supraorbital neuroma, or simple hematoma.6
Multiple imaging modalities can be employed to assess for and characterize an STA pseudoaneurysm. Digital subtraction angiography (DSA) has been the historical gold standard imaging technique; however, DSA only evaluates the patent aneurysmal component, resulting in underestimation of pseudoaneurysmal size in the setting of partial thrombosis. Moreover, DSA is an invasive procedure bearing a small but significant risk of stroke.7 Therefore, initial radiological workup more commonly entails noninvasive imaging studies, particularly duplex Doppler ultrasonography and CT angiography.
While standard, grayscale ultrasound evaluation of a pseudoaneurysm can reveal a cystic, hypoechoic bulging structure abutting a blood vessel, this finding is not specific and can also be associated with simple hematomas, as well as simple or complex cysts.8 Thus, more conclusive diagnosis frequently requires color and spectral Doppler analyses. Color Doppler ultrasonography characterizes directional blood flow by measuring the relative frequency of sound waves emanating from blood flowing within a region of interest. Arterial blood flow directed toward the ultrasound probe will be represented with an orange-red color, while blood flowing away from the probe will be blue, owing to the lower relative frequency of its associated sound waves.9 In the setting of a pseudoaneurysm, bidirectional internal flow within the cystic-appearing pseudoaneurysmal space results in a “swirling” pattern of red and blue signal, reminiscent of the yin-yang Chinese philosophical symbol.
Much like the classic B-mode, grayscale findings mentioned above, the “yin-yang sign” is not necessarily specific to pseudoaneurysm: noncommunicating hematomas that have not fully thrombosed may also display internal flow, owing to force transmission from pulsations within the adjacent artery.10 Thus, conclusive diagnosis requires spectral Doppler analysis, which results in a graphical display of local flow velocity. In the setting of a pseudoaneurysm, spectral Doppler over the neck of the pseudoaneurysm will reveal a “to-and-fro” signal, in which systolic positive flow velocity (ie, flow into the pseudoaneurysmal sac) alternates with diastolic negative flow velocity back into the now relatively low-pressure artery.
Once diagnosed, management of STA pseudoaneurysm is guided by both patient symptoms and risk of rupture. In the case described above, the patient had no complaint of headache or visual deficits (potential sequelae owing to frequent STA anastomosis with the supraorbital artery, which supplies the superior rectus and levator palpebrae superioris muscles), and his current anticoagulation status supported medical management (ie, discontinuing apixaban to promote thrombosis) over surgical intervention. Persistent or rapidly growing pseudoaneurysms or patients presenting with focal symptoms may require more invasive treatment, including vessel ligation or surgical resection of the lesion.11 Successful manual compression has also been reported.12 Regardless of treatment approach, resolution must be achieved to prevent pseudoaneurysm rupture, and this can be confirmed noninvasively using either CT angiography or Doppler ultrasound showing absence of pseudoaneurysmal patency and blood flow.
— Brad Kincaid, MS, MD, is a 4th year diagnostic radiology resident at the Wayne State University in Detroit.
— Nicholas Arconti, BA, is a 4th year medical student at the University of Connecticut School of Medicine in Farmington, Connecticut.
— Mark Kane, MD, is an assistant professor of radiology at UConn Health at the University of Connecticut.
