TIME IS BRAIN
History and Course of Illness
A 57-year-old woman presented to the emergency department within three hours of developing sudden onset left hemiparesis and hemiplegia. A detailed neurological examination revealed profound neglect and right gaze preference. A noncontrast CT scan of the head was performed, followed by CT perfusion (CTP) imaging. Results of both examinations were reviewed by the neurointerventional service, and the patient was subsequently admitted for emergent cerebral angiography and tailored therapy.
Findings
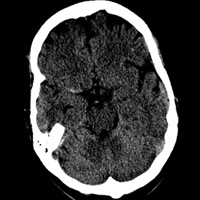
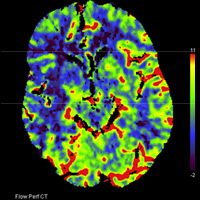
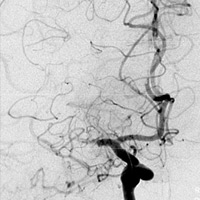
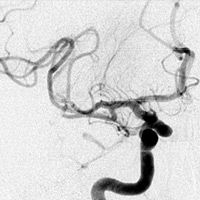
Noncontrast CT demonstrated hyperdense thrombus occluding the right M1 segment (Figure A). There was no evidence of intracranial hemorrhage, mass lesion, or apparent cytotoxic edema. Following the administration of iodinated contrast material, rapid sequential imaging (bolus tracking) of the brain was performed through preselected slices within the middle cerebral artery (MCA) territory. Absolute values of cerebral blood volume (CBV), cerebral blood flow (CBF), and time to peak (TTP) were acquired. Parametric maps of CBV, CBF, and TTP were generated and evaluated qualitatively. TTP was elevated within the right MCA territory (Figure B). A nearly equivalent distribution of decreased CBF was noted (Figure C). Only a focal zone of decreased CBV was observed within the subinsular region, diagnostic for perfusion mismatch (Figure D). Angiography confirmed the presence of occlusive thromboembolus within the proximal right M1 segment (Figures E, F). Intra-arterial (IA) thrombolysis with tissue plasminogen activator (tPA) and direct mechanical thrombectomy utilizing the Penumbra System (PS) were performed, ultimately recanalizing multiple MCA segments (Figure G).
Diagnosis
Acute right MCA territory infarction
Discussion
In the setting of an acute cerebrovascular accident, imaging plays a fundamental role in patient triage and management. Depending on institutional preference and expertise, a variety of advanced imaging modalities can be employed to accurately assess and characterize an acute ischemic event. Initial noncontrast CT examination (for stroke therapy) serves several purposes, namely excluding intracranial hemorrhage, mass, and large territorial infarction.
The principal role of perfusion imaging is to ascertain whether there is “salvageable” brain that can be recovered during a period of acute ischemia. IV administration of tPA is far more common than IA administration, which in a survey conducted between 1999 and 2001 comprised only one-tenth of 1% of patients who received thrombolysis despite the fact that IA therapy is arguably superior. Besides extending the window of treatment from three to six hours, greater accuracy, a higher concentration of tPA at the clot, and increased rates of recanalization result in comparatively better outcomes. Caveats include the requirement for highly specialized physicians and increased rates of intracranial hemorrhage.1
An alternative and/or complementary method of endovascular therapy entails direct mechanical removal of occlusive thrombus. The PS has a larger therapeutic window of eight hours and was designed to disrupt and aspirate thrombus. Recently, a modification of the traditional PS has been devised, referred to as forced-suction thrombectomy. Preliminary data present the modified PS technique as a viable option for acute stroke management.2
Understanding perfusion imaging requires an appreciation of the central volume principle. CBF is determined by the ratio of CBV divided by the mean transit time (MTT). In clinical practice, a surrogate value for MTT that is often used is TTP. Normal brain values of CBF range between 45 and 100 mL/100 g of tissue/minute. Approximately 20mL/100 g/minute is defined as the ischemic threshold at which there is cessation of action potential generation. Irreversible neuronal damage (infarction) typically occurs at values equal to or lesser than 10 mL/100 g/minute.
During an acute ischemic event, MTT (TTP) increases first and represents an area of diminished/slow blood transit, encompassing both infracted and ischemic brain tissue. To maximize oxygen extraction, the inherent autoregulatory response of the brain is to vasodilate vasculature distal to the occlusion or stenosis. Vasodilatation serves to increase CBV so that CBF remains above the threshold for irreversible damage. Beyond this point, any further increase in MTT will ultimately reduce CBV and CBF, respectively.
The concept of the ischemic penumbra is critical to deciding on thrombolytic therapy. It is the penumbra, or tissue at risk, that will benefit from thrombolytic therapy. In fact, a large area of irreversibly damaged brain is a major risk factor for hemorrhagic conversion if tPA is administered. Core infarct size is typically represented by regions of diminished CBV and CBF (less than 10 mL/100 g/minute), which demonstrate abnormal restricted diffusion (DWI+) upon MRI. The region of brain that demonstrates decreased CBF (10 to 20 mL/100 g/minute) and normal/elevated CBV secondary to autoregulation is defined as the ischemic penumbra. As long as the mismatch between core infarct size and the penumbra is greater than 20%, the patient meets the perfusion criteria for thrombolytic therapy.3,4
Despite the therapeutic implications of CT perfusion imaging, issues regarding radiation safety and dose reduction are paramount. In 2009, more than 200 patients received radiation doses well beyond acceptable levels while undergoing brain perfusion CT at one facility. Technical factors (ie, incorrect console settings) were apparently to blame. The FDA notification that followed justifiably heightened national awareness (and contempt). CT quality assurance programs that incorporate reviews of protocols, phantom testing, and actual dose monitoring received by patients are essential to any practice. Radiologists, technologists, and physicists must work together to regulate dose parameters. As physicians and patient advocates, radiologists bear the ultimate burden in deciding the necessity of advanced imaging, implementing methods to reduce radiation dose, and avoiding patient overexposure.5
—Rahul V. Pawar, MD, DABR, is an attending neuroradiologist at Saint Barnabas Medical Center in Livingston, N.J.
 |
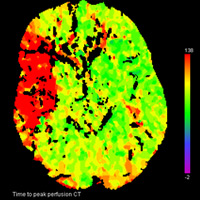 |
| Figure A — Initial noncontrast CT head hyperdense thrombus within right M1 segment | Figure B — Perfusion imaging time to peak |
 |
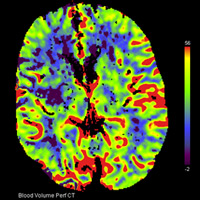 |
| Figure C — Perfusion imaging cerebral blood flow | Figure D — Perfusion imaging cerebral blood volume |
 |
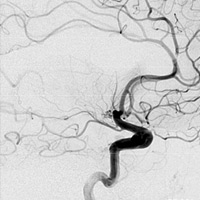 |
| Figure E — Cerebral angiogram, AP view | Figure F — Cerebral angiogram, lateral view |
 |
|
| Figure G — Cerebral angiogram (s/p intra-arterial thrombolysis and thrombectomy), AP view |
REFERENCES
- Qureshi AI, Suri M, Nasar A, et al. Thrombolysis for ischemic stroke in the United States: Data from National Hospital Discharge Survey 1999-2001. Neurosurgery. 2005;57(4):647-654.
- Kang DH, Hwang YH, Kim YS, Park S, Kwon O, Jung C. Direct thrombus retrieval using the reperfusion catheter of the penumbra system: Forced-suction thrombectomy in acute ischemic stroke. AJNR Am J Neuroradiol. 2011;32(2):283-287.
- Nabavi DG, Cenic A, Craen RA, et al. CT assessment of cerebral perfusion: Experimental validation and initial clinical experience. Radiology. 1999;213(1):141-149.
- Yousem DM, Grossman RI. Neuroradiology: The Requisites. 3rd ed. Philadelphia: Mosby; 2010:104-117.
- Wintermark M, Lev MH. FDA investigates the safety of brain perfusion CT. AJNR Am J Neuroradiol. 2010;31(1):2-3.
ON THE CASE submission requirements
- Cases should have clinical relevance and clear radiological findings.
- Sections should include a title, history and course of illness, findings, diagnosis, and discussion.
- Maximum word limit should not exceed 800. At least three references are recommended.
- Cases may be submitted from any radiological subspecialty and imaging modality.
- Figures must be high-quality JPEG or TIFF images and labeled for ease of reference. Please keep images in their native format, without the addition of arrows or other means of highlighting the key findings.